E-Waste Will Be a Global Critical Material Equalizer
Ongoing research makes the prospect of mineral extraction from waste an increasingly viable proposition

Recent articles propelled a conversation with a journalist from a publication that delivers actionable intelligence on global financial markets and the companies and industries that make up those markets. The discussion centered around metals associated with energy transition and the challenges therein. If the journalist’s article is published (and I’m quoted), I’ll link it here. Here’s the background research that formed my opinions as well as my conviction on the role recycling has in the rare earth minerals ecosystem.
A recent article that discussed the impact of China’s (then-upcoming/now-enforced) chipmaking metal export regulations highlighted a key factor which seemingly underpinned Chinese government actions: it held the largest share of the means of production of critical material. Another key element in a recent article about a major company in South Korea’s EV battery sector highlighted how an increasing number of companies are seeking vertical integration with mining sources for this very purpose, i.e. to secure the means of supply of minerals to maintain uninterrupted operations.
Consumption trends of sophisticated electronic goods belie an important fact: countries predominantly in the Western Hemisphere have been generating an outsized amount of electronic waste (or “e-waste”) per capita for some time now — with Norway, Iceland, the U.S. and Canada estimated as being the largest per capita producers.
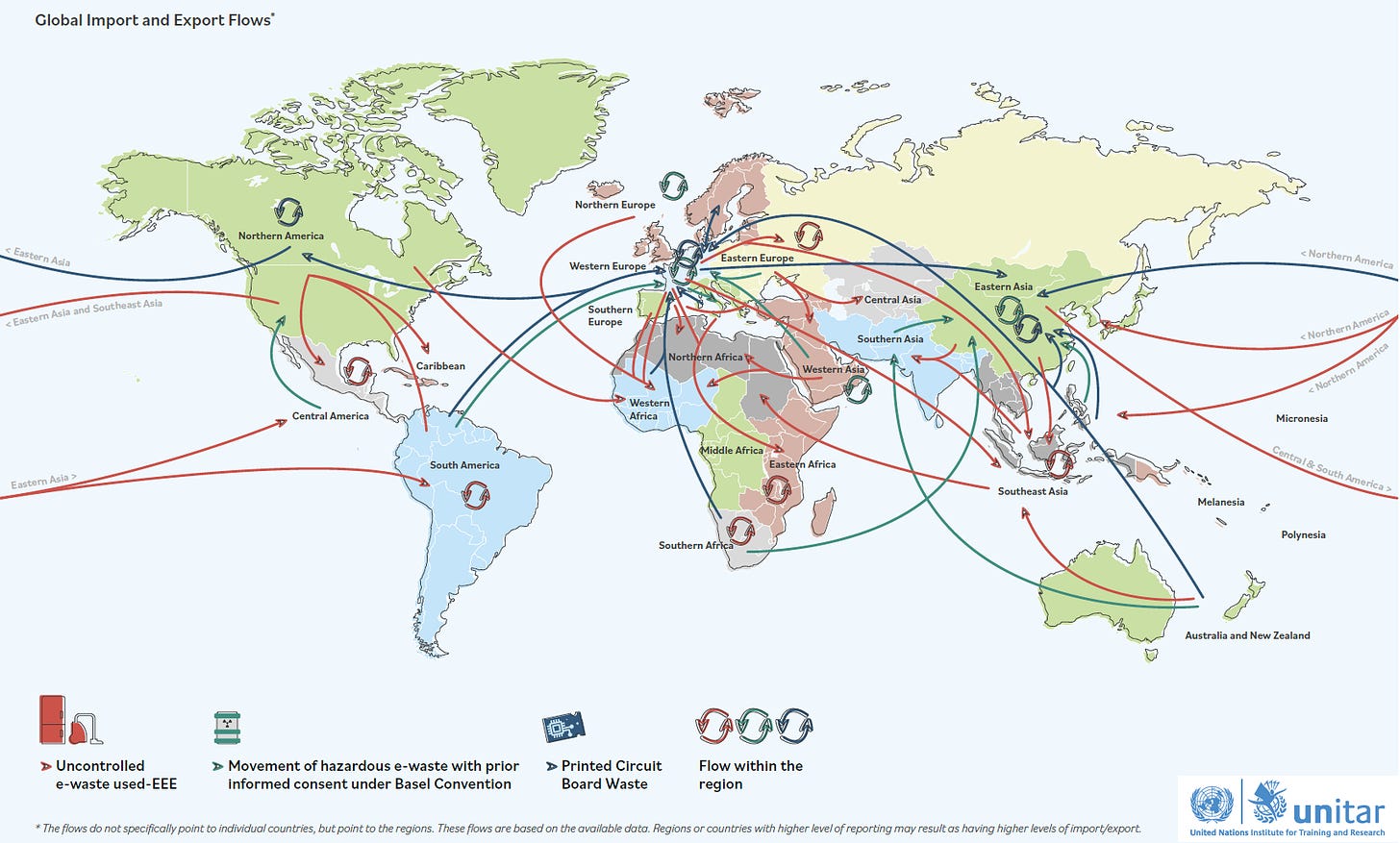
However, none of these countries are the dominant repositories of e-waste. The Global Transboundary E-waste Flows Monitor published for the United Nations Institute for Training and Research (UNITAR) indicates that emerging economies are the dominant recipients of both regulated and unregulated electronic and electric equipment (EEE).
Now, both India and China have increasingly strong e-waste import regulations in place, which seem to imply that some level of treatment of e-wastes are executed prior to being re-exported into these countries for further processing. However, it is estimated that nations of Southeast Asia such as Vietnam, Thailand and Malaysia are relatively more vulnerable than the rest. Overall, it is estimated that:
While Europe, East Asia, and North America do have some capacity to manage hazardous waste and printed circuit board waste (and in fact import them), the recycling capacity for hazardous e-waste is unknown.
Europe, East Asia, and North America are also primary exporters of uncontrolled used-EEE and e-waste exports, mainly to Africa, Southeast Asia, Central America, and South America.
Recipient countries in Africa, Southeast Asia, Central America, and South America have low recycling rates — with North Africa and West Africa being the primary importing hubs for uncontrolled used-EEE and e-waste exports, mostly from Europe.
In 2019, the world generated 53.6 million tons (Mt) of e-waste — an average of 7.3 kg per capita. E-waste generation is expected to increase to 74.7 Mt in 2030 as consumption trends in emerging economies and less developed countries (LDCs) rise. Central to the increasing “energy transition” towards non-polluting technologies have been the increasing utilization of Rare Earth Elements, of which nearly 25% of the REEs Neodymium (“Nd”), Praseodymium (“Pr”), Dysprosium (“Dy”) and Terbium (“Tb”) is devoted to the production of permanent magnets. A typical Electric Vehicle’s motor/generator requires about 2–5 kg of magnets, of which the REE can comprise about 1 kg. The manufacturing of direct drive generators for wind energy requires an estimated average material intensity of 580.9 kg of neodymium magnet (NdFeB or “Neodymium-Iron-Boron”) per MW. Even consumer electronics such as Hard Disk Drives typically have 10–16 g of magnets, which corresponds to about 4 g of REEs.
Until recently, recycling from end-of-life (EOL) consumer products or industrial wastes had been minimal, largely due to the difficult economics. Recent developments in recent years have created new approaches tailored for recovering valuable REEs from EOL products or secondary sources such as permanent magnets from hard disk drives (HDDs), actuators such as low-profile speakers in TVs and mobile devices, compact motor/generators such as the traction motors of electric and hybrid vehicles, industrial wastes including swarfs (machining debris) and breakages from magnet manufacturing, high volume spent catalysts, and so forth.
One notable advantage recycling technologies have is that they avoid the REE “balance problem” wherein the least valuable lanthanides — cerium and lanthanum —constitute the majority of the REEs in ores such as bastnäsite and monazite. With the desired mix already available in EOL products and other forms of feedstock, there is potential for these new technologies to create viable profit opportunities in countries that have hitherto found utility only as dumping grounds for wealthier nations.
Rising Prevalence of Organics for Technology
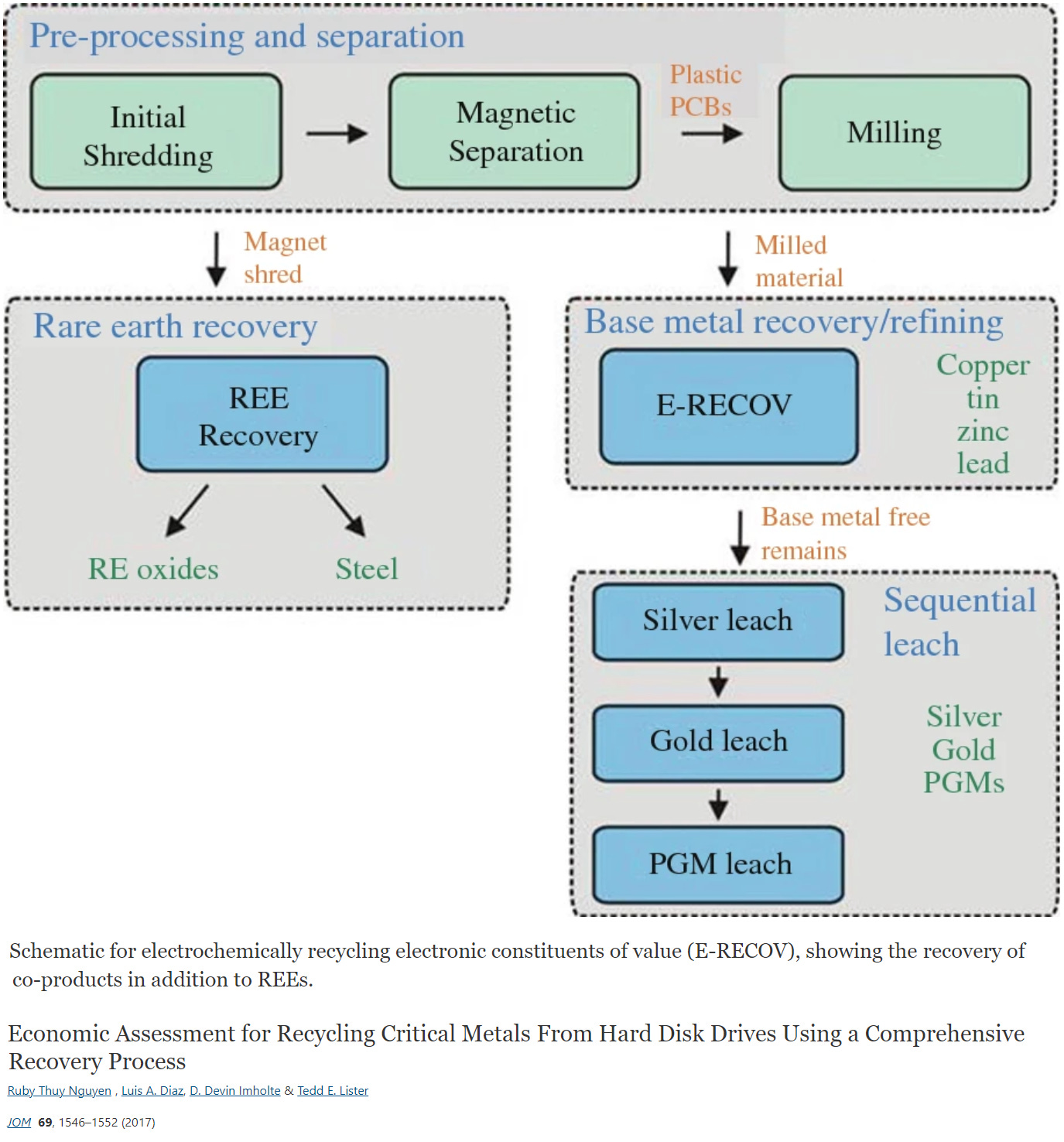
In order to make REE recovery economically viable, contemporary research suggests coupling the recovery of the REEs from the ferromagnetic fraction with processing of the non-magnetic fraction to recover base metals as well as precious metals separately. For instance, it has been posited that shredding feedstock and then pursuing separation allows for a highly effective separation of constituents.
Following this, a number of increasingly innovative methods are currently under development. One such approach is the use of a column of immobilized lanmodulin, a bacterial protein shown to have extremely high affinity for lanthanides, thus facilitating the effective separation of light lanthanides (Nd and Pr) from the higher-value heavy rare-earth dysprosium. If non-REEs are present in the influent, they pass through the lanmodulin column, while the REEs are retained by selective binding to the lanmodulin. Then, because the lanmodulin has higher affinity for light REEs (LREEs) than for heavy REEs (HREEs), the LREEs can be separated from HREEs by sequential stripping at different pH or citrate concentrations.
The deployment of a natural protein is particularly appealing since the protein is biodegradable. However, the economic competitiveness of this approach will hinge in part on the stability of lanmodulin during prolonged use under realistic industrial conditions. Studies of the robustness of the protein are ongoing, as are preliminary techno-economic analyses.
The economics using organics are particularly encouraging. For instance, another approach involved using Gluconobacter bacteria, which naturally produces organic acids that can pull REEs such as lanthanum and cerium from spent catalysts used in petroleum refining or from fluorescent phosphors used in lighting. Despite this bacterial action recovering only about a quarter to half the REEs available within the feedstock, the economics laid out indicate profitability is quite possible. In a hypothetical plant recycling 19,000 metric tons of used catalyst a year, the team estimated annual revenues to be roughly $1.75 million. In a scenario in which the bacteria are fed refined sugar, total costs for producing the REEs are roughly $1.6 million a year, thus leaving around $150,000 in profits. Switching from sugar to corn stalks, husks and other harvest leftovers, however, would slash costs by about $500,000, raising profits to about $650,000.
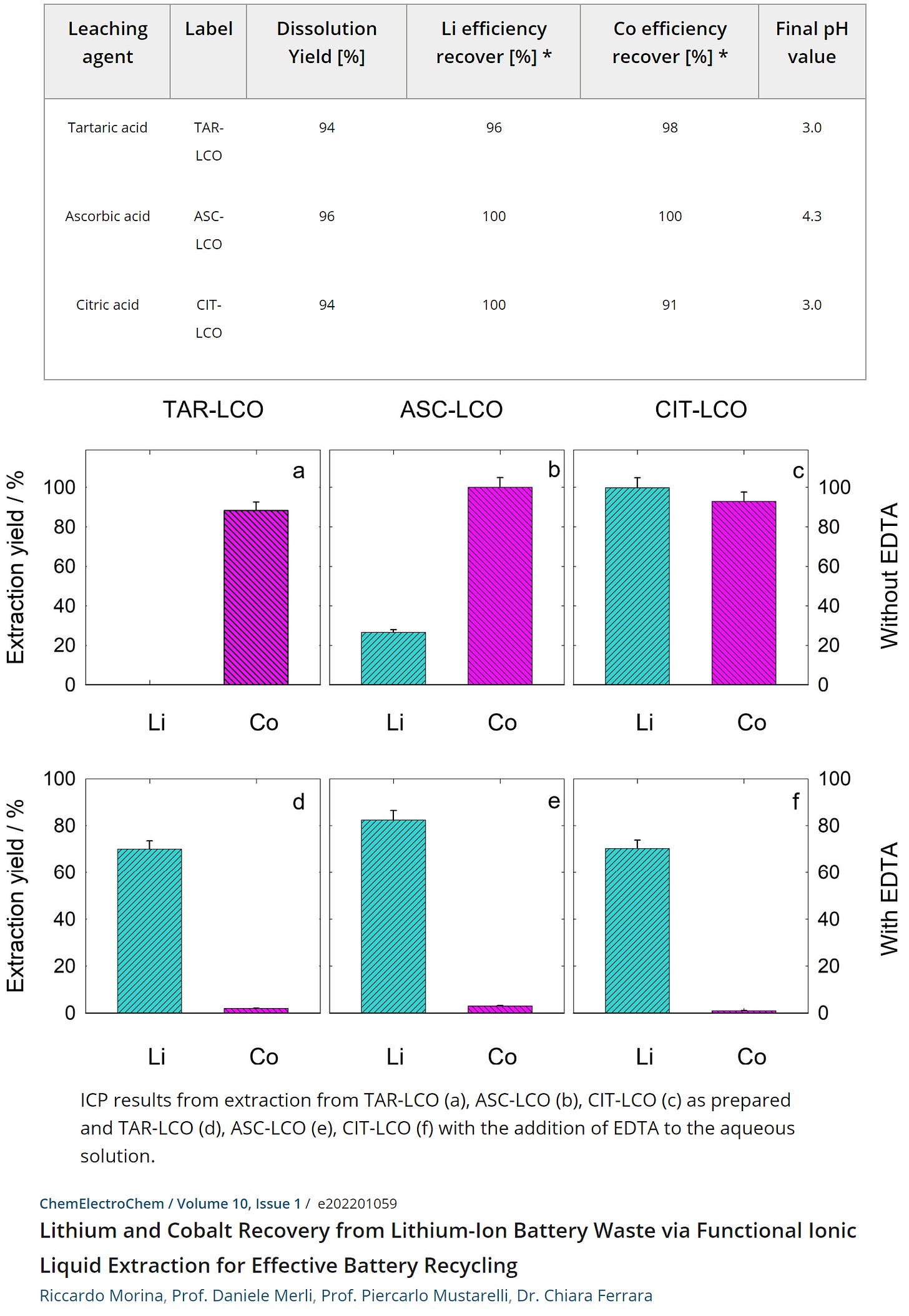
The value of organic acids that tend to be readily biodegradable isn’t limited to the extraction of REEs: a number of ongoing research projects have been showing steady success in extraction efficiency for nickel and cobalt as well. For instance, one such endeavour reported that an ionic liquid — 3-methyl-1-octylimidazolium thenoyltrifluoroacetone or Omim-TTA — can be employed for the selective recovery of lithium and cobalt from the leached solution of a Lithium Cobalt Oxide (LCO) cathode derived from EOL batteries while employing organic acids such as tartaric, ascorbic, and citric acids. The reported efficiency has been promising.
The ionic liquid itself could be fully recovered via simple washing with Hydrochloric Acid (HCl) solution, thus allowing for its reuse in subsequent extraction cycles. Synthetic tartaric acid can be made chemically using maleic anhydrates or hydrogen peroxide (both biodegradable) while citric acid is available in synthetic form as MCA, 99% of which is manufactured using the fungus Aspergillus niger.
Once again, effective industrial deployment requires improvements in stability and turnaround time — a work in progress that will likely show results in the near future.
The likes of nickel has already seen high recycling potential. As early as 2010, the Nickel Institute has reported a recycling rate of 68% from consumer products.
Achieving Economies of Scale
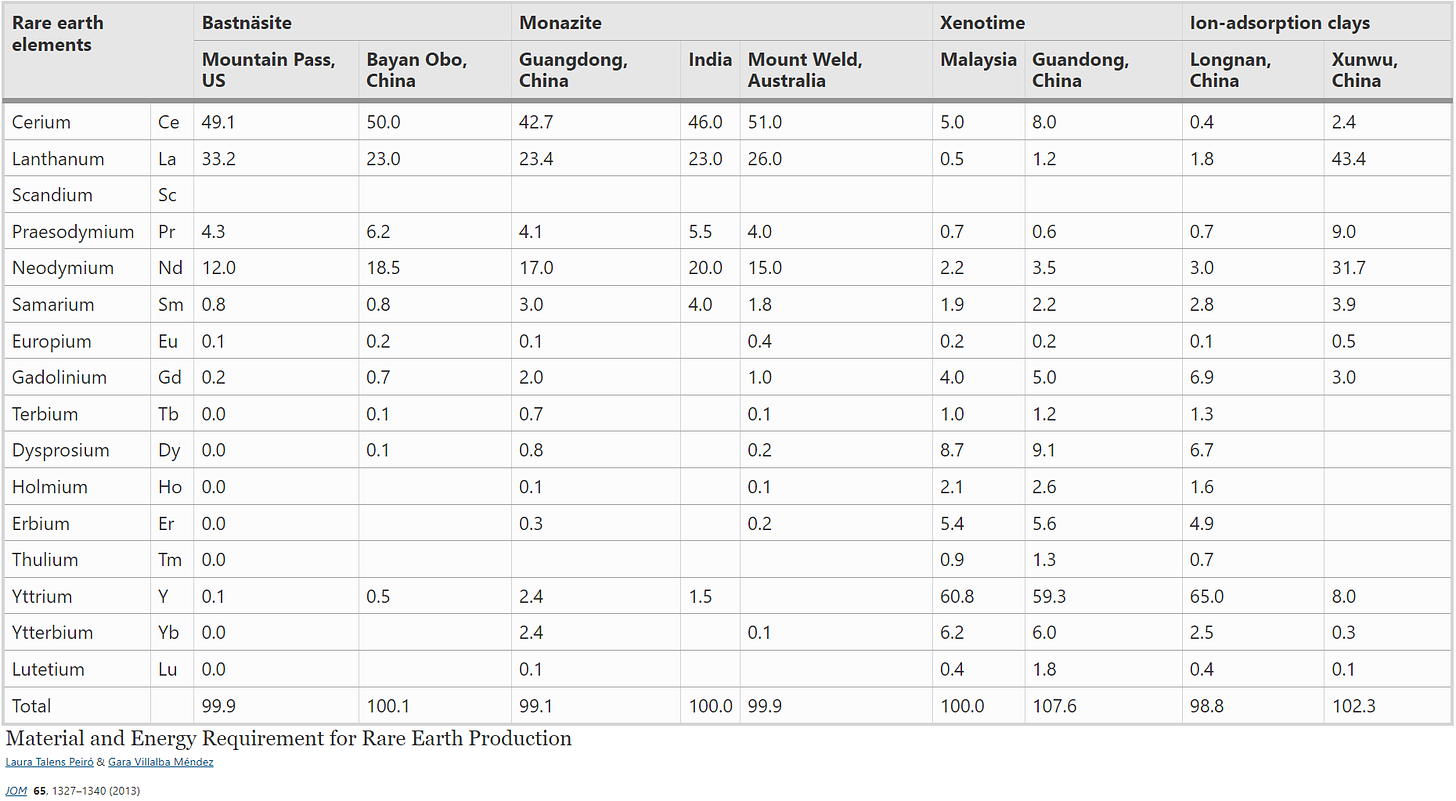
Most REEs are usually found together and mostly in three major minerals: (a) bastnäsite with iron (Bayan Obo, China), (b) monazite and xenotime ores with radioactive thorium (Mountain View, California and Kerala, India), and (c) ion-adsorption clays. Although some European deposits are already identified, for example, in Kvanefield (Greenland) and Norra Kärr (Sweden) in the form of eudialyte, very limited mining activities have occurred thus far. More than 95% of REEs are estimated to occur in bastnäsite, monazite, and xenotime. Bastnäsite contains about 70–75% rare earth oxides (REOs), monazite 65–70%, and xenotime 61–67%. Bastnäsite is the least problematic REO source since it contains very little radioactive thorium.
Although all rare earth minerals are generally open-pit mined, the energy requirement for crushing and grinding varies depending on their hardness and the gangue minerals associated. Typical hard rock ores are mined from the surface by open-pit methods. The ore is blasted, loaded into trucks and transported to a mill. Then, it is processed by crushing and grinding until at least 90% of the particles are no bigger than 0.15 mm.
In Mountain Pass, the primary ore is crushed and turned into a slurry, which is then shipped to plants in China, where individual REEs are separated before being refined into metals and forged into (e.g.) magnets. It is assumed that exhaust gas scrubbing is only installed in modern large-scale plants in China. With minimal to no exhaust gas scrubbing, a significant release of Hydrogen Fluoride (HF) because of acid roasting and other emissions can be expected. However, no plant-specific Chinese data is publicly available.
As of 2013, the estimated energy input for processing 1 ton of REMs varies depending on its concentration in the primary ore mined. At Mountain Pass, the mined ore contains an average of 7.7% of REO and the recovery rate is about 90% due to a very fine grinding of the minerals. Thus, 17 tons of mineral ores and an energy input of 1.74 Gigajoules (GJ) are needed to extract of 1 ton of REM. At Bayan Obo, the mineral ore contains 4.1% of REO and the average recovery rate is 50%; consequently, the extraction of 1 ton of REM requires mining almost 50 tons of mineral ore and 6 GJ of energy input. Bayan Obo is estimated to have improved its recovery rate since then.
The estimates on energy consumption and recovery rate alone puts a boundary case for the making recycling technology viable. If made viable, the burden on the metals industry shifts from feeding total demand to only incremental demand as consumption increases if recycling technology can feed most (if not all) replacement demand. Advancements made by researchers, while still ongoing, is a rather potent “white pill” for the prospects of the metals industry. Furthermore, countries treated as dumping grounds till date will be served an equalizer of sorts via access to an attractive and monetizable foothold within the critical materials ecosystem.
Thus, it will be increasingly important to establish effective policies and incentives to further encourage and propagate recycling technologies around the world.
For a “Big Read” on other matters, the ever-popular “Dharma” series traces the evolution of Eastern faith and philosophy. Here’s Part 1, Part 2, Part 3, Part 4 and Part 5, followed by the ancillary Part 6 and Part 7 discussing Malaysia’s and Indonesia’s spiritual history. Also, click here for a list of all articles published.